What exactly is electricity, and why does it travel through things?
People use 'electricity' casually in their daily lives, but if you think about it, you don't know how electricity flows. Electronic circuit engineer Lucamtukh explains the question, 'What is electricity in the first place, and why do certain materials 'conduct' electricity?'
But good sir, what is electricity? - lcamtuf's thing
https://lcamtuf.substack.com/p/but-good-sir-what-is-electricity

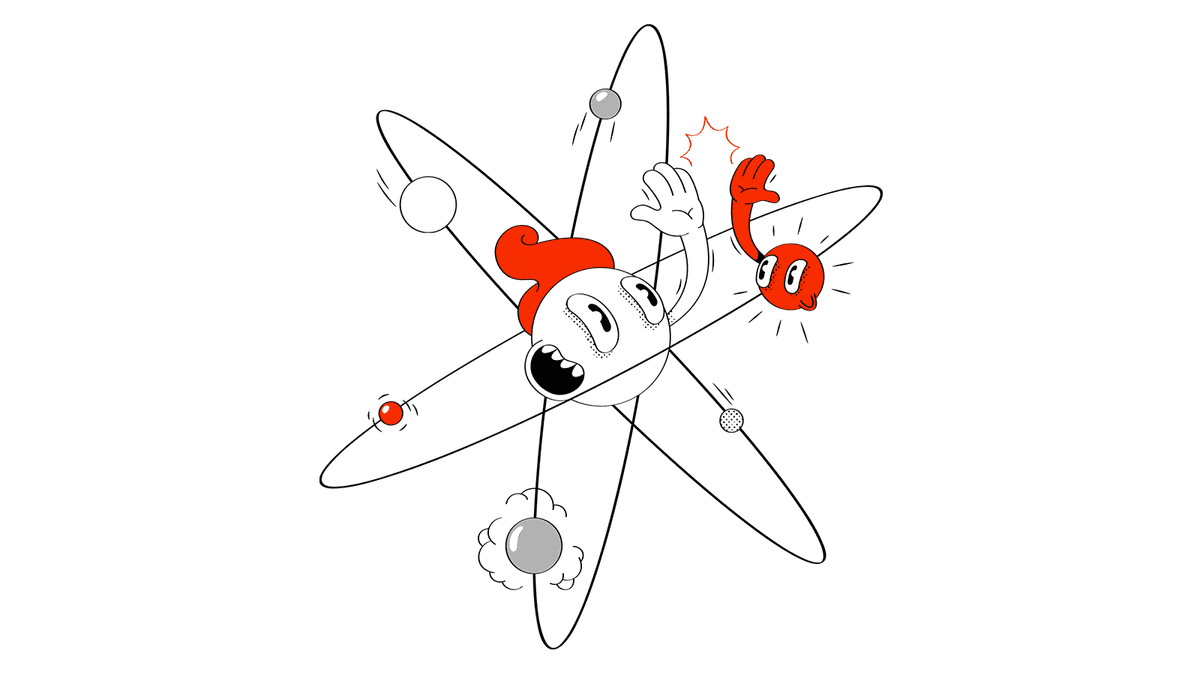
When we learn about atoms at school, we are usually given a schematic diagram like the one below, which shows electrons moving around a nucleus made of neutrons and protons.
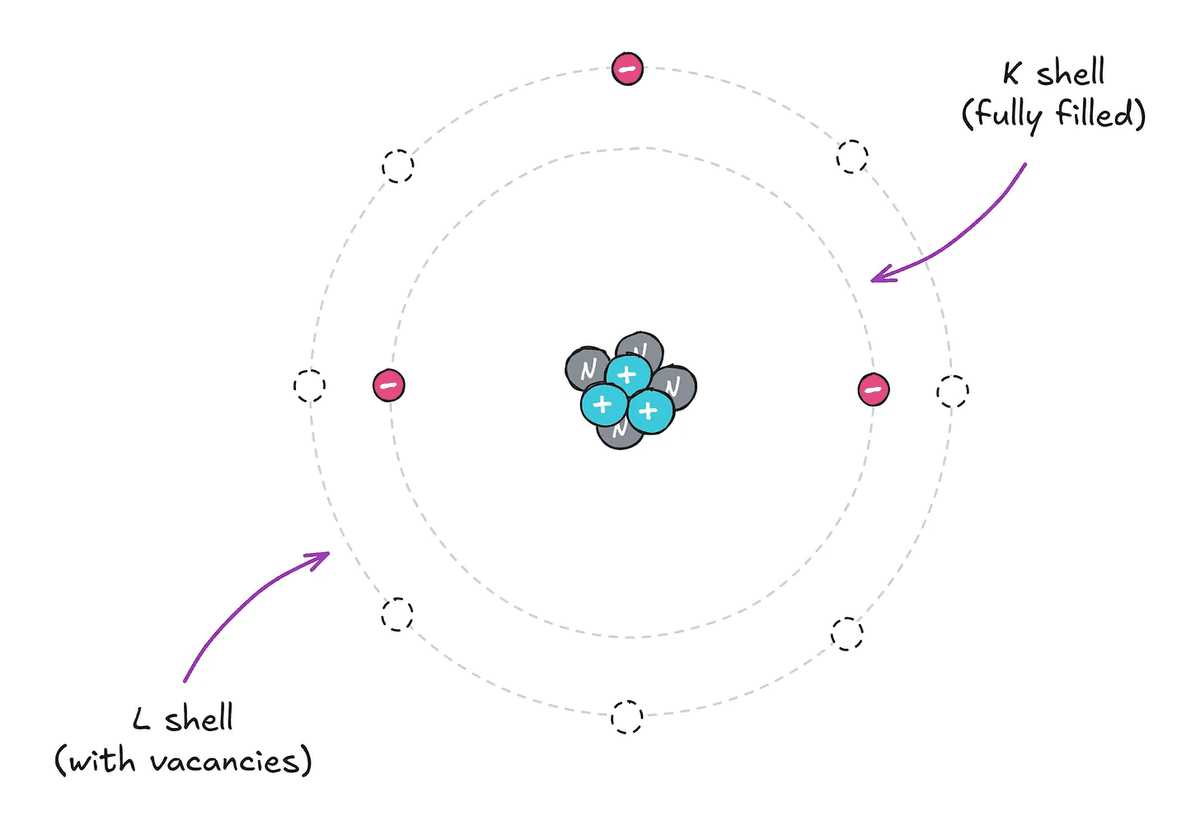
While the diagram above is intuitive and easy to understand, electrons do not actually move in circular orbits, but exist in a specific distribution in quantum mechanical space. However, Lukhamtuk says, 'For now, all you need to understand is that each atom or molecule has a certain number of electrons, and they are housed in a certain number of shells.'
The flow of electricity is closely related to the movement of electrons, but the electrons in the innermost shell of an atom are stable and are unlikely to affect or be affected by the outside world. However, electrons in the outer shell cause various interactions with the outside world.
Chemical bonds that join atoms together to form molecules are also examples of interactions. In covalent bonds, atoms stick together to create a new 'orbital' and some of their electrons move into the new orbital. In ionic bonds, electrons move from one atom to another and the resulting electrostatic field holds the atoms together to form molecules.
It is not difficult to temporarily manipulate some of the electrons in an atom, and this phenomenon is often observed in households as 'static electricity.' When two different substances are brought together, the electrons can move randomly. When the substances are separated, static electricity occurs if the balance of electrons is disturbed. Electrons can easily be pulled away from or deposited on the surface of a substance, but they cannot move around unless there are vacancies in nearby molecules.
In metals, atoms form a dense, homogeneous lattice, which allows the electrons to fuse together and spread throughout the material. This allows the electrons to move like a free-flowing gas. If you remove an electron from a metal and insert it back in, electrostatic forces (Coulomb force) will move the neighboring electron, and then the neighboring electron, and so on, until the entire metal is filled with electrons. This flow of electrons is electricity.
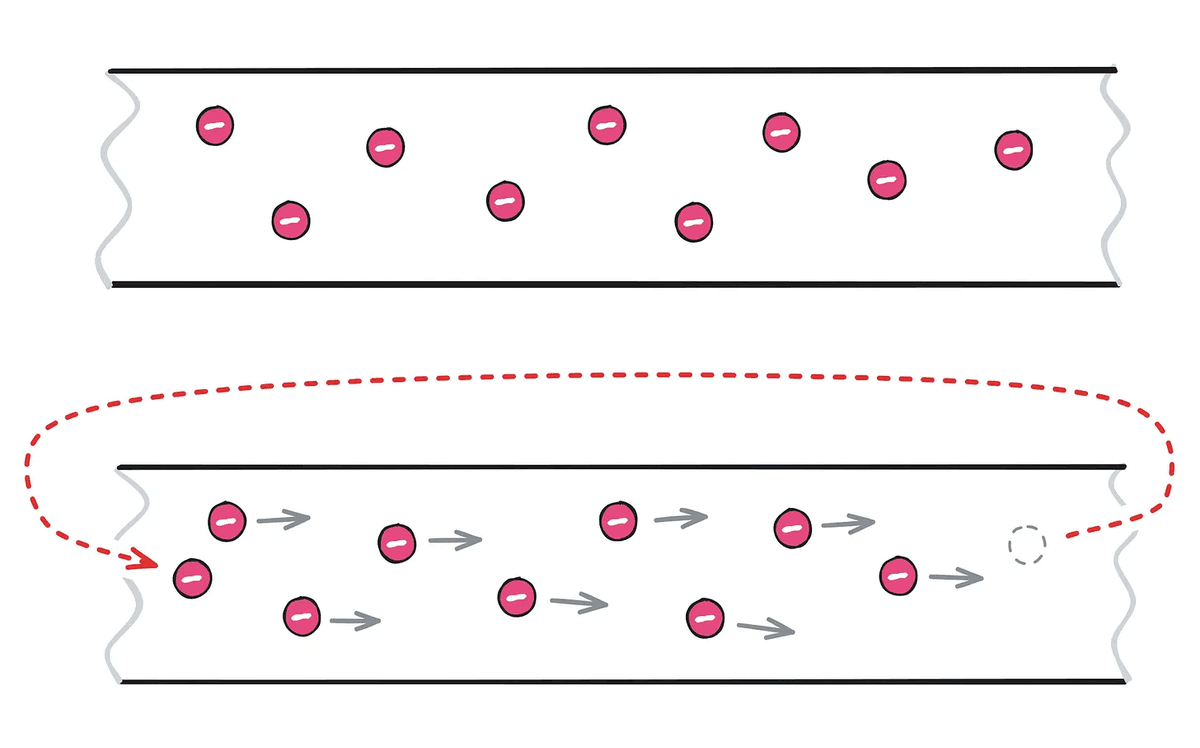
The speed at which electrons move is extremely fast, at about 300,000 km per second, but the movement of each electron in a metal is extremely slow, moving less than 1 cm per hour. Regarding this difference, Lukhamtov gives the example of 'When you shout at someone, your voice travels very quickly, but the air molecules at the shouter's location reach the listener much more slowly.'
There are other mechanisms for the transmission of electricity, such as the movement of electrons due to thermionic emission in a vacuum, as well as the movement of ions in plasmas and highly polar solvents. However, 'it is unlikely that these will be used in modern electronic circuits,' said Lukhamtov.
Related Posts:
in Free Member, Science, Posted by log1d_ts